@Agathe-Zhu
2015-08-15T07:19:38.000000Z
字数 3671
阅读 2224
Secret of Life - Week 2 (Lecture 3-5)
MOOC Biology Course_note
Lecture 3. Protein and Protein Structure
1. Protein: Primary Structure
H2N-CH(R)-CO[OH H]2N-CH(R')-COO == H2N-CHR-CO-NH-CHR'-COOH
* NH2:Amino
* R: Side chain
* COO: Carboxyl
* CO-NH: Peptide bond
Double or more peptide bonds cannot Rotate freely(partial double bonds character)
2. Category of amino acides
20 amino acides into 3 groups
a. Polar, Uncharged
| R | Name | Short for three | Short for one |
|---|---|---|---|
| -CH2OH | Serine | Ser | S |
| -CH(CH3)OH | Threonine | Thr | T |
| CHO-NH2 | Asparagine | Asn | N |
| CH2-CHO-NH2 | Glutamine | Glu | Q |
b. Polar, (-)charged at pH 7
| R | Name | Short for three | Short for one |
|---|---|---|---|
| CH2-COO | Aspartic acid | Asp | D |
| CH2-CH2-COO | Glutamic acid | Glu | E |
c. Polar, (+)charged at pH 7
| R | Name | Short for three | Short for one |
|---|---|---|---|
| (CH2)4-NH3 | Lysine | Lys | K |
| (CH2)3-NH(NH2)+-NH2 | Arginine | Arg | R |
| Histidine | His | H |
d. Hydrophobic
| R | Name | Short for three | Short for one |
|---|---|---|---|
| -CH3 | Alanine | Ala | A |
| -CH(CH3)2 | Valine | Val | V |
| -CH2-CH2-S-CH3 | Methionine | Met | M |
| CH2-CH(CH3)2 | Leucine | Leu | L |
| CH(CH3)-CH2-CH3 | Isoleucine | Ile | I |
| -CH2-Phenyl | Phenylalamine | Phe | F |
| -CH2-Phenyl-OH | Tyrosine | Tyr | Y |
| Tryptophan | Trp | W |
e. Special
| R | Name | Short for three | Short for one |
|---|---|---|---|
| -H | Glycine | Gly | G |
| -CH2-SH | Cysteine | Cys | C |
| Ring | Proline | Pro | P |
-S=S-: Disulfide bond
Problem of protein folding:
1. Varied possiblities of amino acid composition
2. Proteins changes according to environment (not physically/chemically perfect)
3. Proteins' Secondary structure
The proteins' secondary structure was found by neglecting the side chains by Linus Paulin.
Alpha Helix: 3.7 amino acids per turn
Beta Sheets
Loops
Alpha helix and Beta sheets are stablized by hydrogen bonds between C=O and N-H of the polypeptide.
4. Proteins' Tertiary and Quarternary structure
Tertiary structure: Alpha helix and Beta sheets combine together
Quaternary structure: Different proteins combine together by certain bonds like hydrogen bonds formed by Cystein (S=S)
Lecture 4. Enzymes and other amazing machines
1. Design a Protein
| Object | Solution | Explanation |
|---|---|---|
| Situated in the outer membrane of bacteria | Beta Barrel | Non-polar parts inside to get contact with phospholipid tails |
| Make through 600-unit Molecules | Hole | |
| Make through non-charged molecules | (+)&(-) charged amino acides | Get away charged molecules |
2. Enzyme: Triose Phosphate Isomerase (TIM)
| Reactant | Intermediate | Product | ||
|---|---|---|---|---|
| Glyceraldehyde-3-Phosphate | Cis-enediol | Dihydroxylacetone phosphate | ||
| G3P | DHAP | |||
| CHO-CHOH-CH2-PO32- | => | COH=CO-CH2-PO32- | => | CH2OH-CO-CH2-PO32- |
| High free energy | Extremely high | Low |
So enzyme helps getting through the energy obstacle from G3P to cis-enediol by changing activation barrier.
Target of TIM:
1. Stablize the trasition step
- His95 (+ charged) give one H+ to oxygen in the end and grab another H+ from hydroxy
- Glu165 (- charged) grab H from the second Carbon
2. Prevent losing the phosphate
- Hydrogen bonds stablize the phosphate in the end by loop
Limitation: Diffusion of substrate => catalyse effeciently => Perfect
Lecture 5. Biochemical Pathways
1. Energetic reaction
Reaction depends on:
1. Free energy
2. Concentration of composition
For reaction A --> B:
R: Constant which depends on temperature
T: Temperature in Kelvin
if:
2. Logical tricks of pathways
If A --> B is unfavorable, there may be two tricks:
1. Direct coupled reactions, ex: coupled to th ehydrolysis of ATP (ATP --> ADP,
2. Indirect coupled reaction, ex: A--> B --> C
3. Glycolysis
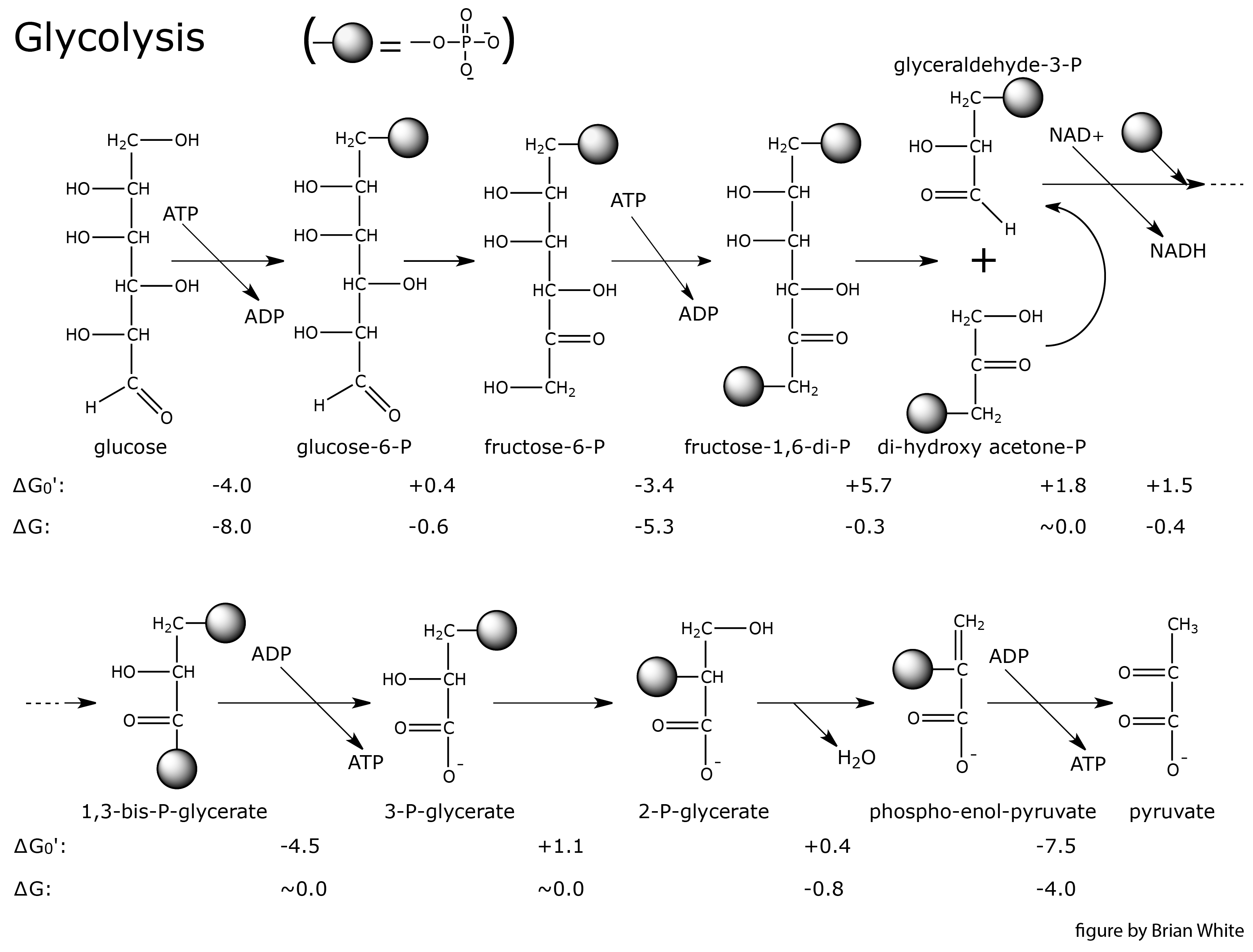
4. Reaction Regulation
- Feedback inhibition, ex: product combines with enzyme witch activates precedent reactions(Allosteric regulation)
- Feddforward activation, ex: substrate combines with activate enzymes
